 Both the emission of a positron and the capture of an electron, a neutrino is always emitted in order to conserve energy. 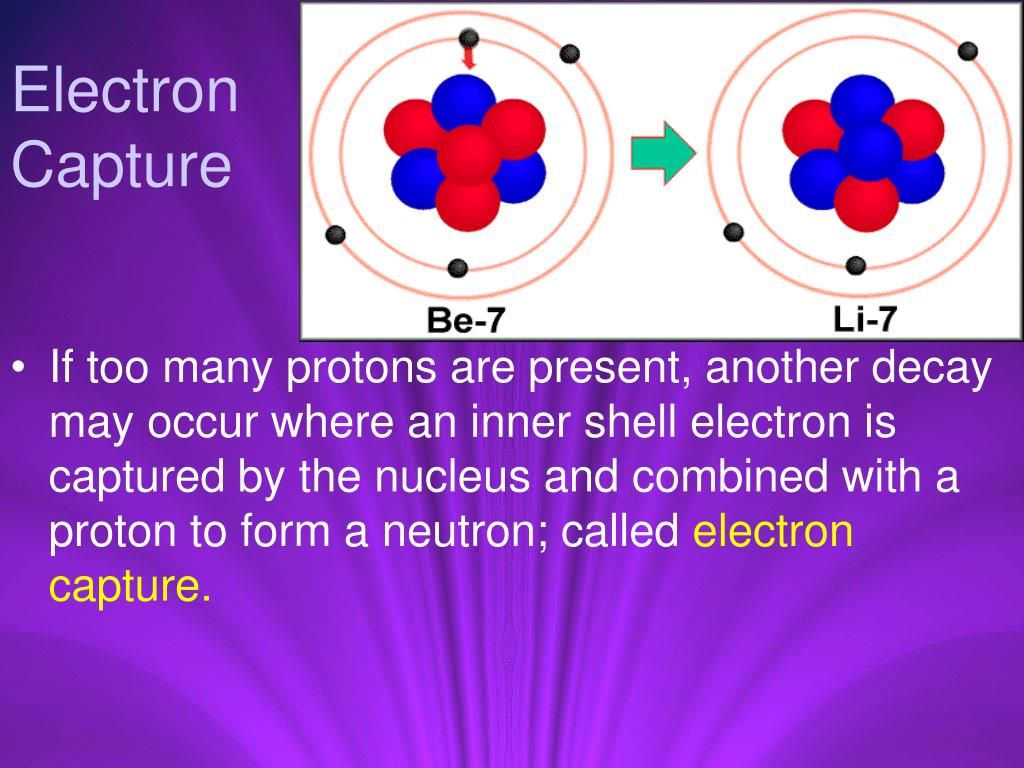
Nevertheless, e + p ( n, e), is a perfectly good. Since a positron cannot for a bound state with a nucleus-it can't be captured. Like the notation used to indicate isotopes, the upper left superscript in the symbol for a particle gives the mass number, which is the total number of protons and neutrons. Electron capture and positron decay are normally competing processes through which a neutron deficient nucleus may attain an increased stability. Sodium-22 undergoes electron capture according to the following nuclear equation: 0 1e +22 11 Na 22 10 Ne. Positron capture is not possible, and here is why: The 'capture' part refers to the fact that the electron is taken from its atomic orbit around the nucleus containing the proton with which it reacts. A positron is often designated by \(\beta+\). The atomic number will decrease by one, while the atomic weight does not change. This rare type of emission occurs when a proton is converted to a neutron and a positron in the nucleus, with ejection of the positron. 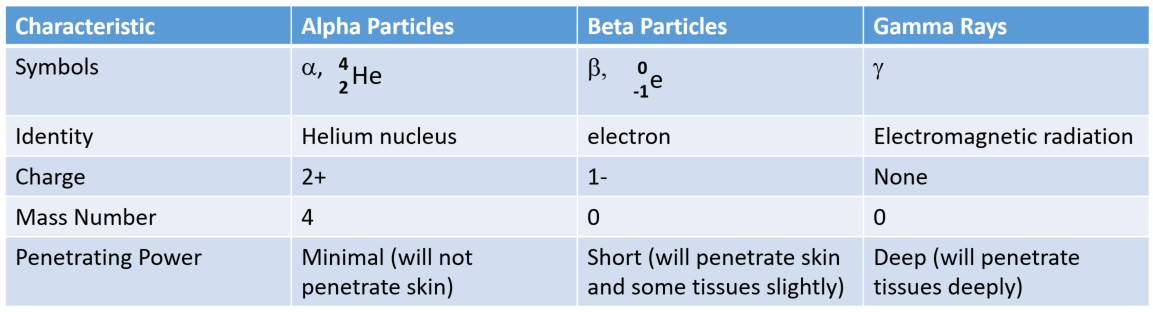
\): Nuclear Decay Emissions and Their Symbols Identity A positron is a positive electron (a form of antimatter). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
